 
In general, the more reactive a metal is: the more vigorous its reactions are the more easily it. Also note on the top right are the activity series for the halogens. The reactivity series of metals is a chart showing metals in order of decreasing reactivity. The alkali metals and heavier alkaline earths on the very top are so reactive that they will displace hydrogen from water, and so are unstable in normal atmospheric conditions. The reactivity series is also known as the activity series of metals. The activity or electromotive series of metals is a listing of the metals in decreasing order of their reactivity with hydrogen-ion sources such as water and acids. 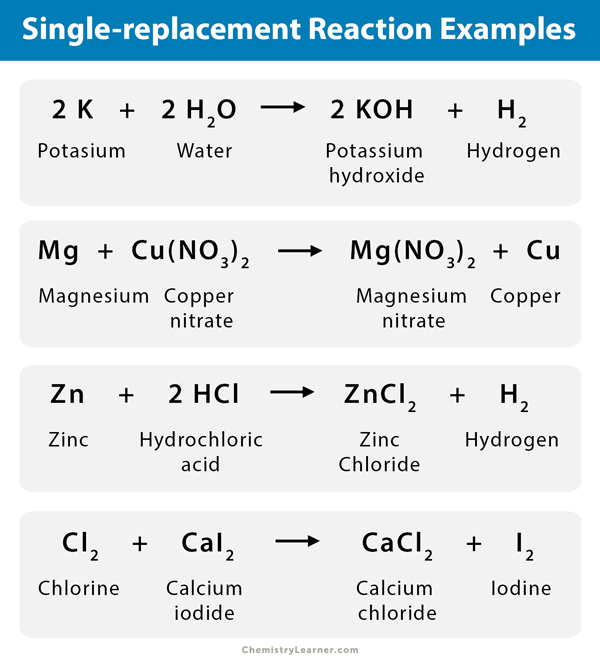
Metals that are more active than hydrogen will displace the hydrogen from an acid. The reactivity series is an ordering of metals from most reactive to least reactive. Explore the examples and uses of the electrochemical. The above table can be used to predict if a single displacement reaction will occur. Christianlly Cena View bio The electrochemical series includes metals ordered by decreasing reactivity or decreasing ease of oxidation. The table summarises some reactions of metals in the reactivity series. \) Activity series of metals and nonmetals reacting in aqueous solutions. The reactivity series tells us if a reaction will happen and under what conditions reactive metal + water metal hydroxide + hydrogen reactive metal + steam metal oxide + hydrogen reactive metal + hydrochloric acid metal chloride + hydrogen Firstly, reactivity affects what a metal can react with. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
